Scaling Cell Therapy: Bottlenecks, Emerging Solutions, and What's Still Needed
By Manish Chamoli, PhD – CSO Partner, LongGame Ventures
Cell therapies can reprogram a patient’s own immune cells to fight cancer or repair damaged tissues, offering cures where conventional drugs fail. The biggest wins so far have been in oncology, especially CAR-T therapies for blood cancers. Now, the broader concept of using living cells as medicine is being applied beyond oncology, into regenerative medicine and aging-related diseases. Stem cell–based approaches are being developed to repair tissues, reset immune function, and target the biology of aging.
Scaling cell therapies isn’t just a technical challenge, it’s the difference between a breakthrough treatment and one that never reaches patients.
Yet despite more than 40 FDA-approved cell therapy products and remarkable clinical results, only ~35,000 patients worldwide have received an approved therapy since the first CAR-T launch in 2017. In the U.S., fewer than 25% of eligible multiple myeloma patients get access.
Why is cell therapy still so hard to scale?
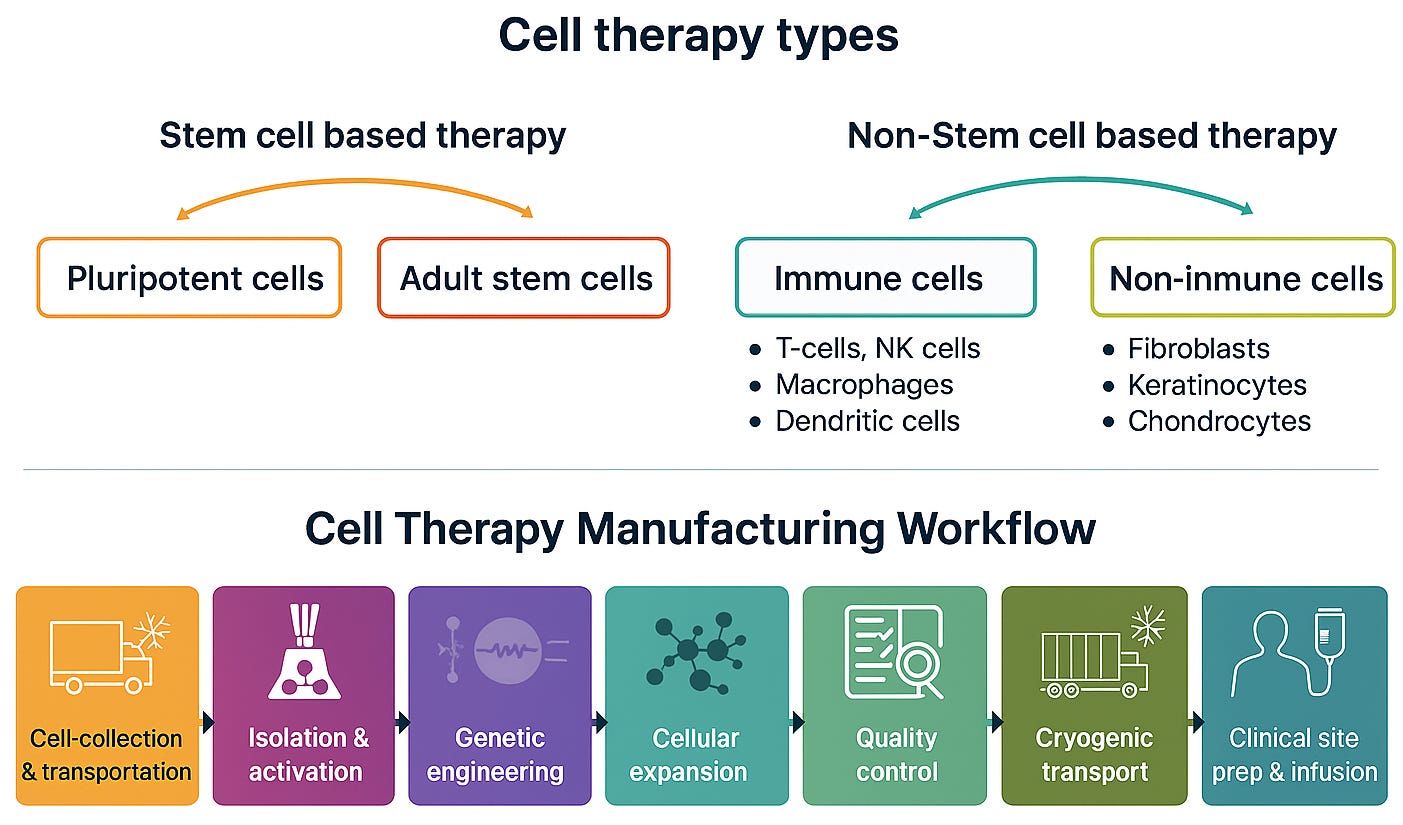
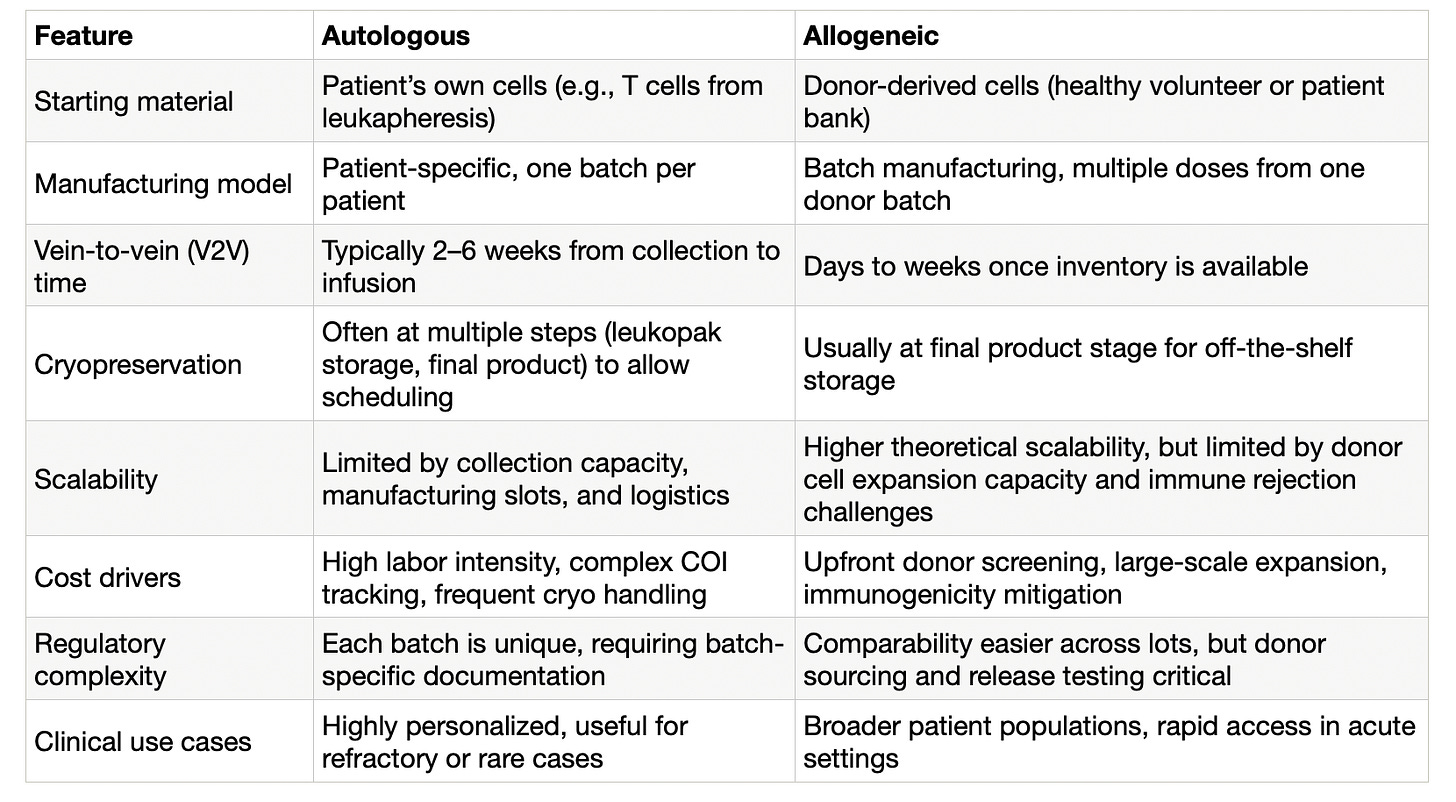
Cell therapies can be autologous (made from a patient’s own cells) or allogeneic (made from donor cells and stored for later use). The “vein-to-vein” (V2V) journey, from cell collection to reinfusion, technically applies only to autologous products, but many of the manufacturing and logistics bottlenecks it reveals are relevant across the field.
Each step of the V2V journey adds technical, logistical, and regulatory friction. These challenges compound into high costs, long turnaround times, and limited patient access.
If we want cell therapies to scale, not just for cancer but also for regenerative medicine and aging, we have to remove these barriers. In this post I will walk through the V2V process to identify key manufacturing, logistics, and regulatory hurdles, and explore technologies that could expand access to cancer, regenerative, and aging-related therapies.
In a future post I will cover the scientific bottlenecks and progress shaping cell therapies for regenerative medicine and aging.
Figure 1: Overview of cell therapy types and manufacturing workflow
Collection: variability starts at the source
Most autologous cell therapies begin with leukapheresis (collecting white blood cells) , using platforms like Spectra Optia (Terumo BCT) and Amicus (Fresenius Kabi), which together account for the largest share of the global leukapheresis equipment market, making them making them the current standard in both clinical trials and commercial manufacturing.
Not all patients yield viable starting material. Chemotherapy can deplete T cells; age, overall health, and mobilization protocols also affect quantity and quality. Some leukopaks fall below viable thresholds, which may require process adjustments or repeat collections.
Once cells are collected, maintaining chain-of-identity (COI) is critical. Any labeling error or routing mistake can lead to an unrecoverable product loss. Digital orchestration tools, such as TrakCel, coordinate COI workflows across multiple collection centers and manufacturing facilities. Many therapies cryopreserve the leukopak for flexible shipping, but this step adds cost and complexity early in the process.
Emerging solutions:
Mobile leukapheresis units, such as the first-in-the-world system launched by BBG Advanced Therapies, bring GMP-compliant collection to underserved or remote areas, expanding access and reducing patient burden.
Closed-system apheresis platforms are increasingly integrating AI-powered monitoring and real-time analytics, which improves process control and consistency across collection, activation, and expansion. This trend is now recognized as a key driver in the growth of the automated cell therapy system market.
Smart cryo-shippers, long used in biopharma logistics, are now becoming standard in cell therapy supply chains. They provide continuous GPS and temperature monitoring, automated alerts, and remote data access, with some platforms adding blockchain-enabled COI tracking to meet the specific demands of living cell products.
What’s still needed:
Collection technology is improving, but variability in leukopak composition from factors such as T cell subset ratios, baseline exhaustion, and total viable counts remains a critical bottleneck that impacts yield, potency, and product consistency. Next-generation platforms could use adaptive closed-loop controls to adjust flow rates, run times, and selection criteria in real time, feeding data directly into GMP records. For e.g., If viable T cells drop mid-collection, the system could extend the run by 15 minutes and log the adjustment automatically. The main hurdle is ensuring these systems are validated, interoperable, and accepted by regulators without adding process complexity.
Table 1: Key differences between autologous and allogenic cell therapy workflows
Isolation & activation: the first gate of control
After collection, the leukopak is enriched for specific cell types, most often T cells. Techniques range from plasticware selection to magnetic bead systems such as CliniMACS (Miltenyi Biotec), one of the most widely used clinical platforms for GMP-compliant enrichment (Ceja et al., 2024). Bead-based isolation improves purity but increases cost due to GMP-grade reagents. Alternatives, including microfluidic and buoyant microbubble-based separations, are being developed to reduce complexity and cost while maintaining yield.
T cells are then activated, typically using anti-CD3/CD28 antibody-coated beads such as Dynabeads (Thermo Fisher) together with cytokines like IL-2 or IL-15. This mimics natural immune stimulation and primes cells for proliferation and engineering. Consistent activation is challenging with patient-derived cells; overactivation can cause functional exhaustion, while underactivation limits yield (Patel et al., 2025)
Closed, automated systems such as CliniMACS Prodigy and Lonza Cocoon are now widely deployed in clinical and expanding commercial workflows, integrating enrichment, activation, and early expansion in sealed GMP environments to reduce contamination risk and operator variability.
Label-free enrichment platforms, including microbubble sorting (Akadeum) and microfluidic closed systems (Astraveus), remain in early-stage adoption but aim to improve reproducibility and reduce cost.
Adaptive activation protocols, combining IL-7/IL-15 with inline metabolic monitoring and AI-guided tuning, are in pilot GMP use. AI is now being applied to optimize stimulation in real time (Pharmaceutical Technology, 2025).
What’s still needed:
Platforms must better integrate real-time functional QC and closed-loop control so manufacturing systems can adapt to patient-to-patient variability without compromising yield or potency.
Genetic engineering: the costliest, riskiest step
Once cells are activated, they are genetically modified to modulate specific genes. This can involve introducing a synthetic CAR receptor, enhancing persistence, reducing immune rejection, or correcting a disease-causing defect.
Lentiviral transduction remains the most widely used method for stable gene addition due to its efficacy but is costly and complex to scale. Production still depends heavily on transient transfection in HEK293 cell lines, which creates variability and cost pressure. Non-viral methods such as electroporation, transposon systems, and CRISPR-based edits offer flexibility and lower raw material costs, but often with trade-offs in efficiency, durability, or regulatory familiarity (Geng et al., 2025).
Emerging solutions:
Suspension-based viral vector production: Manufacturers are shifting from adherent to suspension systems, enabling larger batches and higher yields. Stable producer cell lines and “mega-batch” runs help reduce cost and supply constraints. Hybrid viral–non-viral strategies are emerging to improve flexibility and reduce reliance on a single vector type. (Tridgett et al., 2024; Formas-Oliveira et al., 2025)
Closed-system integrated transduction: Automated, sealed platforms now combine activation, transduction, and early expansion to standardize delivery and cut contamination risk. Proof-of-concept runs have shown shorter timelines and reduced operator input.
AI-guided and real-time QC: AI analytics and inline monitoring can predict transduction efficiency and detect early batch failures, potentially improving yield consistency. Adoption is still early and will depend on regulatory acceptance (Cerbo et al., 2025).
Non-viral delivery alternatives: mRNA transfection, transposon systems, and nanoparticle carriers are in early clinical use. These methods aim to cut costs, shorten timelines, and improve safety, but have yet to match viral vectors for stable, long-term expression in approved products (Moretti et al., 2022).
What’s still needed:
Production systems must deliver consistent potency without sacrificing safety or adaptability. Multiplex editing in workflows to remove TCRs, eliminate HLA, and insert safety switches though promising, also adds complexity, cost, and off-target risk (Moradi et al., 2024). Inline quality control for accuracy, potency, and off-target effects remains a major gap.
Expansion: scaling cell numbers without losing function
Once engineered, cells must be expanded to therapeutic doses, often in the range of 10⁷ to 10⁹ viable cells. Traditional open systems (bags, flasks, G-Rex) require manual handling and feeding, which is labor-intensive and hard to scale.
Rapid expansion depletes nutrients and builds up waste (lactate, CO₂), creating metabolic stress and reducing fitness. Over-expansion risks exhaustion, cutting persistence and potency. Maintaining stem-like T cell phenotypes at scale remains a key challenge.
Emerging solutions:
Now widely deployed: Closed, automated bioreactors like Cytiva WAVE and Sartorius Biostat offer perfusion feeding and environmental control, stabilizing growth conditions and improving consistency.
Growing trend: Automation plus Process Analytical Technology (PAT) is gaining traction. For example e.g., Cellares’ Cell Shuttle uses Raman spectroscopy for real-time monitoring and process feedback, while Terumo BCT’s Quantum Flex captures up to 720 daily data points (glucose, lactate etc.) to forecast expansion trajectories and regulate perfusion.
What’s still needed:
Most platforms still lack adaptive, closed-loop control that adjusts based on live metabolic or potency data. Inline potency assays are rare, and full V2V digital integration remains a goal rather than standard practice. Experimental strategies such as transient telomere extension, cytokine and signaling modulation, metabolic support, or epigenetic reprogramming may help preserve proliferative capacity without compromising safety
Quality Control: reducing delays without compromising safety
Every batch must pass identity, purity, potency, viability, sterility, mycoplasma, endotoxin, and vector copy number checks. Sterility cultures alone can take up to 14 days, locking up inventory and delaying patient treatment. Many QC operations are performed at sites separate from manufacturing facilities, adding transport time, sample integrity risks, and extra layers of documentation.
Emerging solutions:
Inline analytics for real-time monitoring (e.g., Raman and metabolite sensing) to catch problems early and stabilize growth conditions. Examples include MAVERICK style in-line systems and vendor–platform integrations reported for cell therapy bioprocesses.
Rapid sterility approaches using solid-phase cytometry or microfluidics can cut turnaround from days to hours, but require broader validation and regulatory acceptance.
Automated QC workcells that consolidate assays and documentation (e.g., Cell Q) are being deployed, though still limited in number.
What’s still needed:
Move from batch-end, manual testing to real-time, in-process verification that can trigger corrective action before failure. Consensus on rapid sterility and inline potency standards is essential, coupled with digital traceability that connects QC data directly to lot release decisions, shortening release timelines from weeks to days without sacrificing patient safety. (Szarzynski et al., 2025)
Cryopreservation & Logistics: enabling scale, but not without trade-offs
Cryopreservation supports flexible scheduling and global distribution, but at a cost. Most products are frozen at –80 °C or in liquid nitrogen (–196 °C) with 5–10% DMSO, which protects against ice crystal damage but is toxic at higher concentrations. Each freeze–thaw cycle, often done twice per patient can reduce viability and potency, particularly for fragile cell types.
At scale, liquid nitrogen transport adds complexity: validated dry shippers, strict chain-of-custody, and continuous temperature tracking to avoid excursions. These requirements drive cost and operational risk in both autologous and allogeneic workflows.
Emerging solutions:
Novel alternative DMSO-free storage methods such as hydrogel encapsulation or hypothermic (4 °C) preservation are in preclinical stages. Lowering DMSO to ~5% may improve viability and reduce adverse effects. Cryoprotectants like trehalose, sucrose, glycerol, and novel blends are being tested with advanced thawing methods such as magnetic induction heating.
Robotic cryostorage systems to improve throughput and minimize handling errors.
Logistics tech: IoT-enabled shippers with GPS, shock, and temperature sensors for live monitoring; automated dewar replenishment alerts; and blockchain-based chain-of-identity records to reduce transit failures.
What’s still needed:
Validated, clinically accepted DMSO-free cryoprotectants; harmonized global transport standards; and logistics platforms that integrate real-time shipment data with manufacturing and clinical scheduling. Regional manufacturing and point-of-care models, including fresh delivery within 24–48 hours or use of cryo-hubs, could reduce transport distances and improve access, but remain largely pilot-stage due to regulatory and staffing constraints.
Clinical Infusion: the final mile still has friction
Most patients receive lymphodepleting chemotherapy (such as fludarabine and cyclophosphamide) a few days before infusion. If manufacturing is delayed after preconditioning begins, patients may face risk without receiving treatment, making precise coordination essential.
At the clinic, the frozen product must be thawed, verified, and infused according to strict SOPs. Some products require post-thaw dilution or washing, while newer protocols favor simplified “thaw and infuse” workflows. Inconsistent thawing procedures or insufficiently trained staff can reduce the delivered cell dose or introduce variability. Infusion reactions such as Cytokine Release Syndrome require immediate clinical intervention and close monitoring.
Emerging solutions:
Mobile cleanroom units with point-of-care manufacturing services (e.g., Orgenesis OMPUL/POCare™) are being piloted to deploy GMP manufacturing capacity within weeks at hospital sites.
Decentralized “cells-on-demand” systems integrating AI-guided automation and cassette-based workflows are under development, some supported by federal funding, to enable local production with minimal staff, smaller infrastructure footprints, and shorter turnaround times.
Table 2: Scaling cell therapy: Bottlenecks and emerging solutions
Regulation: the quiet constraint on scaling
Even if manufacturing and logistics bottlenecks are solved, the last and often least visible hurdle is regulatory. The system was built for static, synthetic drugs, not for living, patient-specific medicines. Once a cell therapy is approved using a specific set of tools and methods, making improvements can be far more complex than simply upgrading equipment. Changes often trigger costly, time-consuming comparability studies, and in some cases new trials, creating strong disincentives to adopt better, safer, or more scalable processes. As a result, manufacturers may be locked into decades-old methods long after superior technology is available.
Fragmented global rules add further friction. Developers must prepare and submit nearly identical approval packages separately for the U.S., Europe, the U.K., Australia, and other markets. Each jurisdiction applies slightly different requirements, consuming months or even years while patients wait. Efforts within the ICH Cell and Gene Therapy Discussion Group aim to harmonize these standards, but practical implementation remains slow.
For low- and middle-income countries, the barriers are even higher. Regulatory guidelines are often non-standardized or entirely absent, with little precedent for governing cell-based or other advanced therapies. Many regions lack not just legislation, but also the infrastructure, facilities, and technical expertise needed to safely deliver these treatments. Awareness among regulators and clinicians may be limited, and without clear rules, investment and technology transfer stall. In practice, this means the gap in access between high-income and resource-limited settings continues to widen.
There are signs of progress. In the U.S., the FDA’s Advanced Manufacturing Technology designation allows platform-level reviews, so therapies built on the same manufacturing backbone can move through approval more quickly. Project Orbis offers a framework for parallel oncology reviews across multiple countries. At the state level, Montana’s new law allowing access to certain unapproved advanced therapies has drawn attention, though its real impact will depend on adoption and federal alignment.
Ultimately, the field needs a risk-based, globally harmonized approach that enables faster integration of new technologies without compromising safety. Without it, even the most advanced automation, or decentralized manufacturing methods will still collide with a regulatory layer that moves slower than the science. For patients, that means longer waits, even when the production line is ready to deliver.
Summary and looking ahead
Cell therapies have proven their ability to cure cancers and repair damaged tissues where conventional drugs fall short. Yet despite more than 40 FDA approvals and thousands of ongoing trials, patient access remains a fraction of what’s possible. The vein-to-vein journey still contains too many points of friction, from variable starting material and labor-intensive manufacturing, to slow, fragmented regulatory pathways.
Across this workflow, three themes stand out:
Biological variability remains the root challenge. Even the most advanced automation cannot fully compensate for differences in patient-derived cells, donor quality, or immune compatibility.
Integration beats incrementalism. Closed, automated, and data-driven platforms that unite multiple steps, from collection to QC, show the greatest promise for improving yield, consistency, and speed.
Regulation is the ultimate rate-limiter. Without harmonized, adaptive frameworks, technological advances will hit a wall long before their full benefit reaches patients.
The next decade will likely see:
Decentralized manufacturing hubs and point-of-care systems reducing transport delays and improving access in both high-income and underserved regions.
Next-generation preservation and transport technologies that minimize cell damage and simplify logistics.
Modular, AI-guided manufacturing platforms enabling faster process adaptation without triggering full regulatory re-approval.
Broader allogeneic adoption once immune rejection and persistence barriers are addressed, allowing true “off-the-shelf” scalability.
Getting there will require coordinated action:
Industry must invest in scalable, integrated manufacturing platforms with embedded quality control.
Regulators must modernize review pathways to allow safe, rapid incorporation of process improvements.
Healthcare systems must prepare infrastructure and reimbursement models that can deliver cell therapies beyond elite cancer centers.
If we succeed, cell therapy could move from a last-resort treatment for a few thousand patients a year to a routine option for millions, not only in oncology, but across regenerative medicine and aging-related diseases.
— Manish Chamoli, PhD
CSO Partner, LongGame Ventures
Thanks to Will Harborne, Founder & GP, LongGame Ventures, for quick feedback on this piece.`
LongGame Ventures is backing the next generation of companies building scalable cell therapies for age-related diseases. If you’re building an early-stage company addressing bottlenecks in scaling cell therapies, reach out at info@longgame.vc.